Ever seen a headline screaming “Coffee prevents cancer!” or “New research proves turmeric cures everything!” – and then two weeks later, another one says “Coffee increases cancer risk”?
Welcome to the confusing world of health research.
In today’s internet jungle, every influencer waves around a “study” to sell a product. The problem is that most people don’t actually know how to read medical studies or distinguish between scientific evidence and misinformation.
Let’s fix that. You don’t need a PhD or a lab coat. You just need a few simple checkpoints that can help you tell solid evidence-based medicine from fancy-sounding junk science.
Why You Should Learn to Read Medical Studies
Every day, thousands of health articles float around online. Some are honest, but many manipulate data to increase clicks or sales.
A small study on 30 mice becomes a viral post about “miracle fat-burning herbs”.
A preprint (not even peer-reviewed) is quoted on YouTube as if it were the gospel truth.
This is how health misinformation online spreads: by mixing half-truths with scientific language.
Understanding medical research doesn’t mean you must read complex statistical equations. It just means you know what to look for:
- Who did the study?
- Where was it published?
- Was it on humans or hamsters?
- How big was it?
Once you start asking small questions like this, then fake health things on social media won’t fool you so easily.
Let’s go step-by-step.
Step 1 – Check the Source: Where Was the Study Published?
The first red flag is often the source.
If a study appears in peer-reviewed journals like The Lancet, JAMA, Nature Medicine, or NEJM, it’s probably legit. These journals have strict review processes.
But if it’s from a random website or “Global Journal of Superfoods & Longevity,” be careful. Some journals are predatory – they take money to publish anything, no matter how poor the research is.
Search the study title on PubMed, Google Scholar, or a major university site. If it’s not listed there, it’s not serious research.
Also, remember: a press release or preprint is not the final word. Preprints haven’t been peer-reviewed yet. They might later be corrected, withdrawn, or even disproven.
Step 2 – Understand the Study Type
Not all studies mean the same thing. A lot of confusion comes from people not knowing what kind of study they’re reading.
1. Randomized Controlled Trial (RCT)
This is the gold standard. People are randomly assigned to groups – one gets the treatment, the other gets a placebo.
RCTs give the strongest proof for cause and effect.
As Dr. Archibald Cochrane argued, randomized trials are essential – without them, medicine risks being an art, not a science.
If a supplement truly works and has been properly studied, there’ll usually be an RCT showing it. The lack of RCT evidence often means it either hasn’t been studied or doesn’t work well enough to show clear results
2. Observational Study
This one just observes. It can show associations, not causes.
Example: “Coffee drinkers live longer.” Sounds great, but maybe coffee drinkers also exercise more, or earn better income, or sleep better.
So, correlation ≠ causation.
3. Meta-Analysis or Systematic Review
This combines multiple studies to provide a comprehensive conclusion. If done properly, this is one of the most trustworthy forms of scientific evidence.
Step 3 – Who Funded or Conducted the Study?

Always follow the money. Was it funded by a company selling the same product being studied? Then, you know what to expect.
If a supplement company funds a study about its own pill, the chance of a “positive” result magically goes up. Not always dishonest – but bias creeps in.
Look for author disclosures and affiliations. Independent research (done by universities, government, or non-profit institutes) tends to be more reliable.
Step 4 – Pay Attention to the Sample Size and Duration
Here’s something the media rarely tells you: A study on 25 people for two weeks means almost nothing for real-life decisions.
A good study has:
- Enough participants (hundreds or thousands)
- Diverse age, gender, and background
- Long follow-up duration
Small studies are interesting, but cannot be generalized.
Is this study large enough (done on a large population/participants) to apply to me?
Step 5 – Read the Results Carefully

This part confuses most people because of tricky words like “risk reduction.”
If a study says “50% lower risk”, that sounds huge – but what if the actual number went from 2 people in 100 to 1 in 100? That’s just a 1% absolute change.
Always look for:
- Absolute vs relative risk (absolute = the real change in numbers; relative = looks bigger because it’s shown as a percentage)
- Statistical significance (p < 0.05) (A p-value of 0.05 (or 5%) is a commonly used threshold for statistical significance which means researchers are quite sure the result didn’t happen by luck)
- Effect size (is it clinically meaningful?)
And remember – if no one else has replicated the result, treat it as interesting, not the truth.
Step 6 – Correlation Is Not Causation
This one’s the mother of all misunderstandings.
If ice cream sales go up and drowning cases go up, does ice cream cause drowning? Of course not – both increase in summer.
But many health headlines are built exactly like this.
“People who eat spicy food have lower heart disease.”
Maybe those people also eat more vegetables or have active lifestyles.
Always ask: Could another factor explain this result?
Step 7 – Look at Who Was Studied
A study on 20 college students in one city doesn’t apply to everyone.
If the research was on healthy young men, it may not apply to women, the elderly, or people with health conditions.
Before you accept a claim, check if the study population matches you or the person it’s meant for. Understanding medical research includes understanding who it was done on.
Step 8 – Avoid Being Fooled by Media Headlines
This is the trickiest part, where media misinterpretation of studies happens most.
Journalists often simplify or exaggerate for clicks. “Scientists discover secret to long life!” sounds way better than “Preliminary study shows weak association.”
Before you believe any headline:
- Go to the original study or at least the abstract.
- See if the study even said what the headline claims.
- Be wary of words like “miracle,” “breakthrough,” “scientists prove,” or “finally discovered.”
Real science is slow, cautious, and full of “may,” “might,” and “could.”
Step 9 – Cross-Check With Trusted Medical Sources
One study doesn’t make a new medical truth. That’s why organizations like CDC, WHO, Harvard Health, Mayo Clinic, and NIH consistently review the totality of evidence.
If multiple respected sources interpret a finding the same way, that’s your green signal.
If only one influencer is shouting about it while major health bodies stay silent, you can guess why.
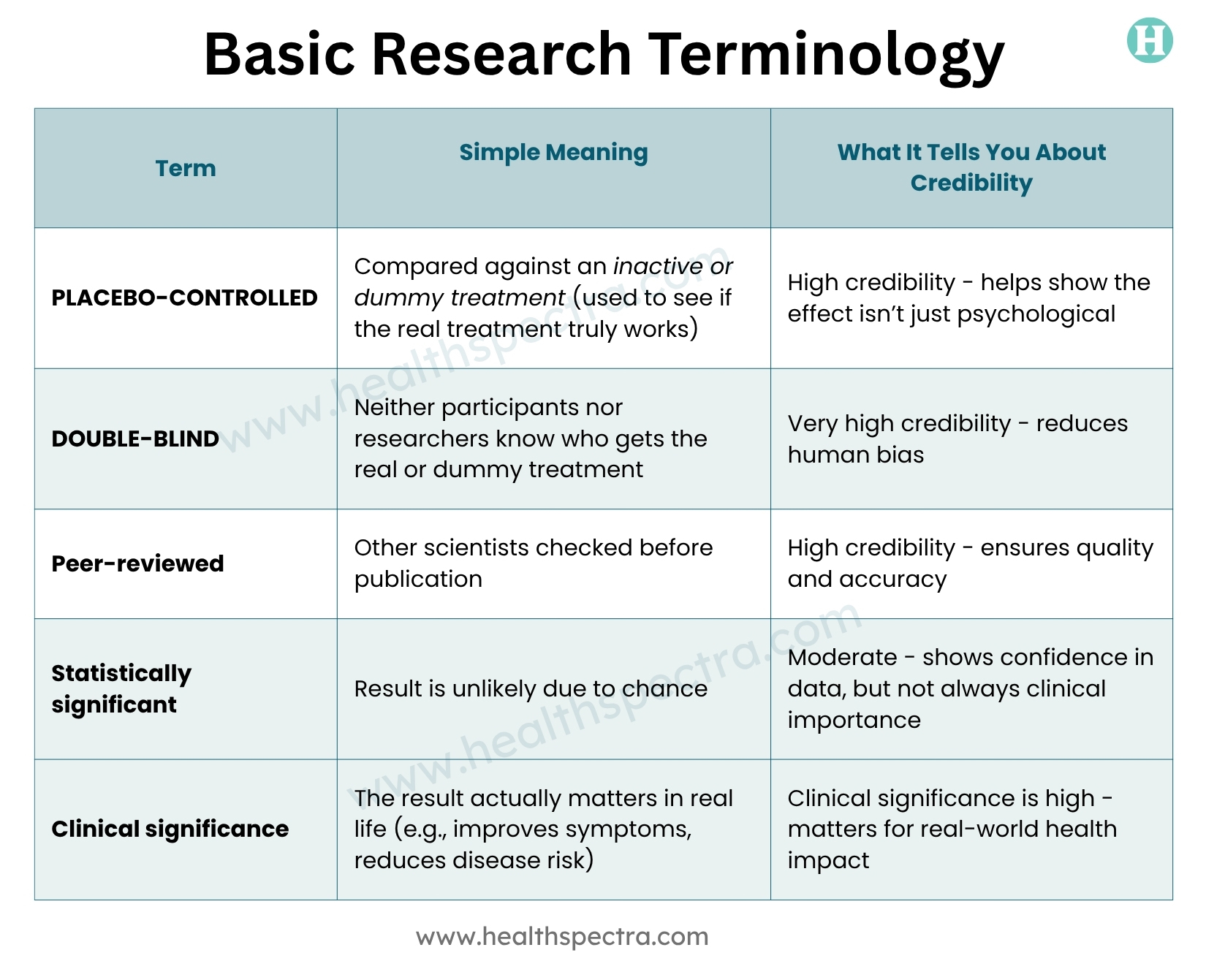
Step 10 – Learn Basic Research Terminology
You don’t need to learn full biostatistics, but knowing a few terms helps a lot. Knowing these words helps you spot pseudoscience that throws around fancy terms without meaning.
Key Takeaway
You don’t need a medical degree to spot fake health claims – you just need curiosity and common sense.
When you see a health claim online, pause and run a quick mental checklist:
- Is it from a peer-reviewed research source?
- What type of study is it?
- Who funded it?
- How big and long was it?
- Does it show causation or just correlation?
- Do trusted medical institutions agree?
If any of these fail, treat it as marketing, not medicine.
Once you start applying this mindset, your scientific literacy grows – and you’ll instantly see how much wellness advice is just clever packaging of weak evidence.
Final Word
In a world full of noise – detox teas, biohacking fads, miracle vitamins – your best defense is critical thinking.
Learning how to interpret scientific studies isn’t about becoming a skeptic; it’s about becoming smarter with your health.
Next time someone says, “Studies prove this works,” don’t nod. Ask:
Which study? Who did it? How many people? What kind of study?
That small pause will save you from believing 90% of the misinformation floating around online.
Read More: Debunking Nutrition Myths for People with Type 1 Diabetes
FAQs About Reading Medical Studies
Q1: How do I know if a study is trustworthy?
A: See if it’s published in a peer-reviewed journal and indexed in PubMed. Avoid random blogs quoting “research” with no link.
Q2: Are preprint studies reliable?
A: Not yet. They’re early drafts – useful for scientists, not for making health decisions.
Q3: How do I find the full study instead of just the headline?
A: Search the title on Google Scholar or PubMed. Sometimes you can find PDFs on university or ResearchGate pages.
Q4: What’s the difference between “statistically significant” and “clinically important”?
A: Statistically significant means the result does not happen by chance. Clinically important means it helps in real life, also, like reducing BP enough so the chances of a heart attack become less.
Q5: Where can I learn how to read, analyse, and interpret a scientific paper ot study?
A: Websites like Cochrane, NIH, or Coursera’s basic biostatistics courses are great starting points. Even free YouTube channels by doctors explain it simply.
References
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3687192/
- https://medlineplus.gov/understandingmedicalresearch.html
- https://pmc.ncbi.nlm.nih.gov/articles/PMC8486420/
- https://www.nia.nih.gov/health/healthy-aging/how-find-reliable-health-information-online
- https://toolkit.ncats.nih.gov/glossary/observational-study/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3687192/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC4296634/
- https://www.ncbi.nlm.nih.gov/books/NBK63647/
- https://www.ncbi.nlm.nih.gov/books/NBK459346/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3444174/
- https://journals.sagepub.com/doi/full/10.1177/2515245917745629
- https://pmc.ncbi.nlm.nih.gov/articles/PMC6910766/
- https://pmc.ncbi.nlm.nih.gov/articles/PMC3505292/
In this Article